Konference: 2014 19th Congress of the European Hematology Association - účast ČR
Kategorie: Maligní lymfomy a leukémie
Téma: Aggressive Non-Hodgkin lymphoma – Clinical
Číslo abstraktu: S1345
Autoři: MD Tadeusz Robak, PhD.; MD Huiqiang Huang, PhD; MD Jie Jin, PhD; MD Jun Zhu; Ting Liu; MD Olga S Samoilova; MD Halyna Pylypenko; Prof. Dr. Gregor Verhoef, Ph.D.; Noppadol Siritanaratkul; Evgenii Osmanov; Julia Alexeeva; Juliana Pereira, PhD; prof. MUDr. Jiří Mayer, CSc.; Xiaonan Hong; Yoshiharu Maeda; Lixia Pei, PhD; Brendan Rooney, PhD; MD Helgi van de Velde, PhD; Prof. MD Franco Cavalli, FRCP
ABSSUB-4224
Background: MCL is an incurable type of aggressive non-Hodgkin lymphoma. Progression-free survival (PFS) is limited with standard frontline therapy (e.g. R-CHOP) in newly diagnosed MCL patients (pts). Bortezomib (V) is approved for relapsed MCL in the US and >50 other countries. Incorporating V into frontline combination therapy may improve outcomes in MCL pts.
Aims: This study determined if adding V in place of vincristine in R-CHOP improved outcomes in newly diagnosed MCL pts unsuitable for bone marrow transplantation (NCT00722137).
Methods: Consenting adults with measurable stage II–IV MCL and ECOG PS 0–2 were randomized 1:1 (stratified by IPI score and disease stage) to 6–8 21-d cycles of rituximab 375 mg/m2, cyclophosphamide 750 mg/m2, doxorubicin 50 mg/m2, all IV d 1, and prednisone 100 mg/m2 PO d 1–5, plus V 1.3 mg/m2 IV d 1, 4, 8, 11 (VR-CAP) or vincristine 1.4 mg/m2 (max 2 mg) IV d 1 (R-CHOP). Primary endpoint was PFS by independent radiology review (IRC). Secondary endpoints were time to progression (TTP), time to next anti-lymphoma therapy (TTNT), overall survival (OS), response by modified IWRC, and safety. 486 pts were planned for 295 PFS events.
Results: Between May 2008 and Dec 2011, 244 pts were randomized to R-CHOP and 243 to VR-CAP; in all 487 pts, median age was 66 yrs, 74% were male, 74% had stage IV MCL, and 54% had an IPI score of ≥3. 97% of pts had MCL diagnosis confirmed centrally. Pts received a median of 6 cycles. After 40 mos’ median follow-up, median PFS by IRC (298 PFS events) was 14.4 vs 24.7 mos with R-CHOP vs VR-CAP (Figure), and by investigator assessment (307 PFS events) was 16.1 vs 30.7 mos (hazard ratio [HR]=.51, P<.001). Median TTP by IRC was 16.1 vs 30.5 mos (R-CHOP vs VR-CAP; HR=.58, P<.001) and by investigator was 16.8 vs 35.0 mos (HR=.47, P<.001); median TTNT was 24.8 vs 44.5 mos (HR=.50, P<.001), and median OS was 56.3 mos vs not yet reached (HR=.80, P=.173; 4-yr OS rates: 54% vs 64%). By IRC, complete response (CR) + unconfirmed CR (CR+CRu) rates (bone marrow and LDH verified) were 42% vs 53% (R-CHOP vs VR-CAP; odds ratio [OR]=1.7, P=.007); by IRC, radiological CR+CRu rates were 71% vs 83% (OR=2.0, P=.002) and overall response rates (ORRs; CR+CRu+partial response [PR]) were 90% vs 92% (OR=1.4, P=.27). By investigator, CR+CRu rates were 28% vs 42% (R-CHOP vs VR-CAP; OR=1.9, P=.002) and ORRs were 92% vs 96% (OR=2.0,P=.07). Median duration of radiological response with R-CHOP vs VR-CAP was 18.1 vs 36.5 mos in pts with CR+CRu+PR by IRC and 16.6 vs 39.2 mos in pts with CR+CRu by IRC. For R-CHOP vs VR-CAP, 85% vs 93% of pts had grade (G) ≥3 adverse events (AEs); those in ≥10% of pts in either arm were neutropenia 67% vs 85%, leukopenia 29% vs 44%, thrombocytopenia 6% vs 57%, lymphopenia 9% vs 28%, anemia 14% vs 15%, and febrile neutropenia 14% vs 15%. Despite the difference in thrombocytopenia rates, rates of bleeding events were similar for R-CHOP vs VR-CAP (any G: 5% vs 6%; G ≥3: 3 pts vs 4 pts). Peripheral neuropathy rates were 29% vs 30% (any G) and 4% vs 8% (G ≥3). With R-CHOP vs VR-CAP, 30% vs 38% of pts had serious AEs, 7% vs 9% discontinued due to AEs, and there were 3% vs 2% on-treatment drug-related deaths.
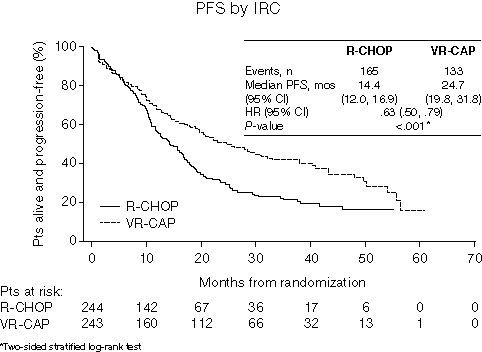
Summary/Conclusion: PFS was significantly prolonged and secondary efficacy endpoints consistently improved with VR-CAP vs R-CHOP, with additional but manageable toxicity, in newly diagnosed MCL pts.
Keywords: Bortezomib, Mantle cell lymphoma, Phase III, Safety
Datum přednesení příspěvku: 15. 6. 2014
